Identifying Cortical Cell Types Through Immunofluorescence and Electrophysiology
Written by Airi Watanabe, Ph.D. Candidate at McGill University
The Cerebral Cortex
The cerebral cortex is the outermost part of the brain, made up of a milieu of neuronal and glia cells. It is responsible for cognitive functions and is involved in memory, attention, and consciousness. Different cortical regions are also associated with specific functions, such as the visual cortex for sight and the somatosensory cortex for sensation. The neocortex, which makes up approximately 90% of the cerebral cortex, is organized into 6 horizontal layers, and neurons are arranged radially in functional cortical columns, as illustrated below.
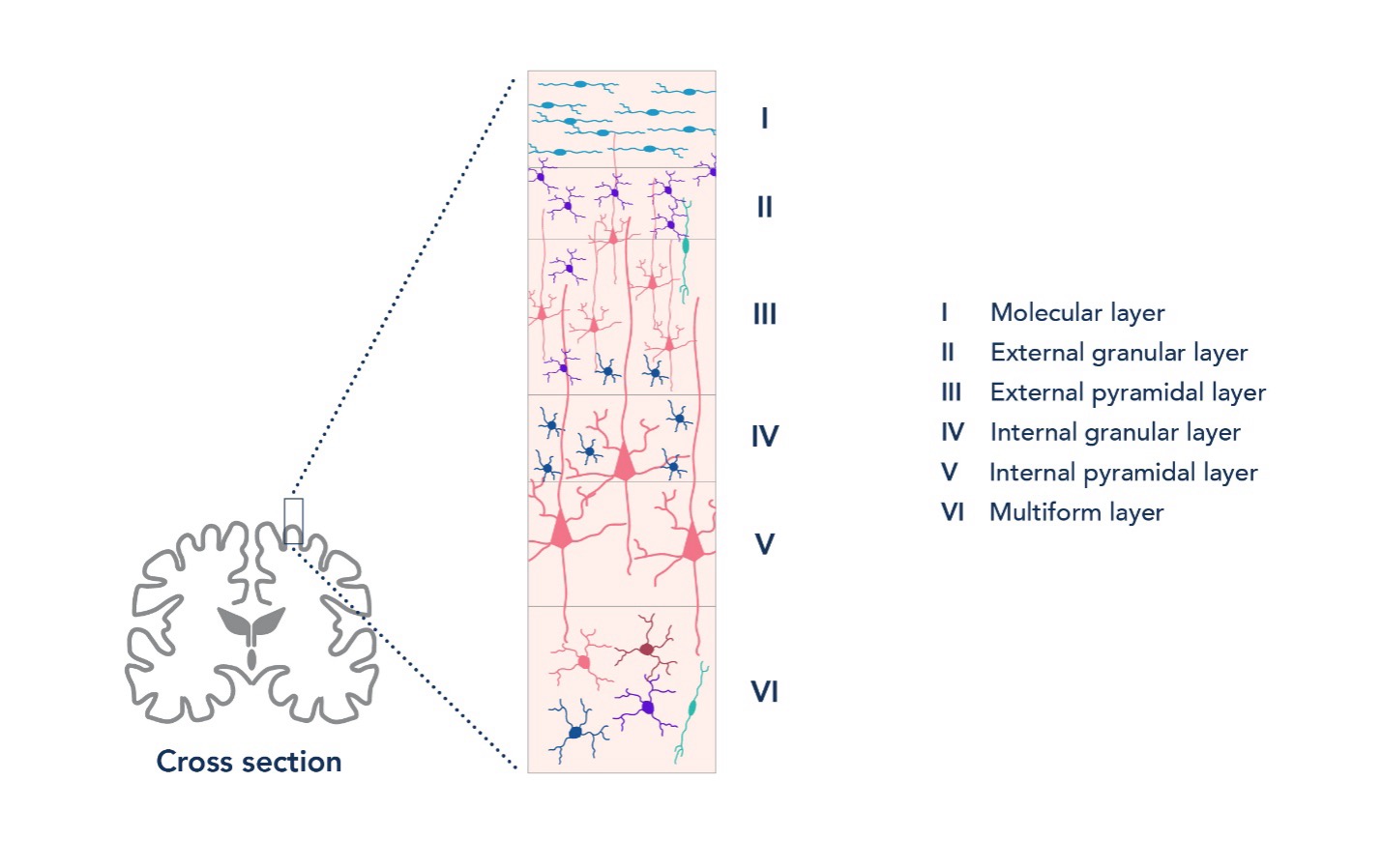
Figure 1. The neocortex is made up of 6 cortical layers, each with distinct compositions of neuron types.
To carry out its specific functions, the cortex is made of circuits that contain many different types of specialized cells. The two main classes of cortical neurons are projection neurons and interneurons. Projection neurons in the cortex are excitatory pyramidal cells, which make up 80% of all cortical neurons. Interneurons are a varied group of cells that mostly have inhibitory functions in the cortex, and these include basket cells and Martinotti cells.
Different neuronal cell types work together to ensure the proper functioning of cortical circuits. Dysfunction in the balance of cortical excitatory and inhibitory cell functioning is a hallmark of neuropathologies, such as epilepsy and schizophrenia. Given the diversity of neuron types, being able to tease apart cell type-specific functions will aid in the development of more precise treatments for neuropathologies. It is therefore important to identify specific neuronal cell types when studying cortical connectivity and function in health and disease.
Introduction to Electrophysiology
In neuroscience, electrophysiology (or “ephys”) is used to measure the electrical activity of living neurons. The patch-clamp technique, pioneered by Bert Sakmann and Erwin Neher, is a powerful method in electrophysiology and is commonly used to study precise changes in the membrane potential of individual cells.
This article will focus on whole-cell patch clamping in acute brain slice. This is where a glass pipette, filled with an internal solution that mimics the intracellular make up of the cell, is used to access the intracellular contents of a cell (1). First, the pipette approaches a cell (2), then a mild suction is applied to the cell membrane to form a seal (3). Once a seal is formed, more suction can be applied to “break through” the membrane to achieve access to the cytoplasm of the cell (4). The internal solution in the pipette can then be dialysed into the cell (5) and this allows the recording and manipulation of membrane potential changes. Other types of patch clamp techniques exist too, such as cell-attached and perforated patch. By measuring the electrical activity of cells, we can, for example, investigate how cellular learning and plasticity take place, how neurological diseases change connectivity in brain cells, and the effects of pharmacological compounds on brain cells.
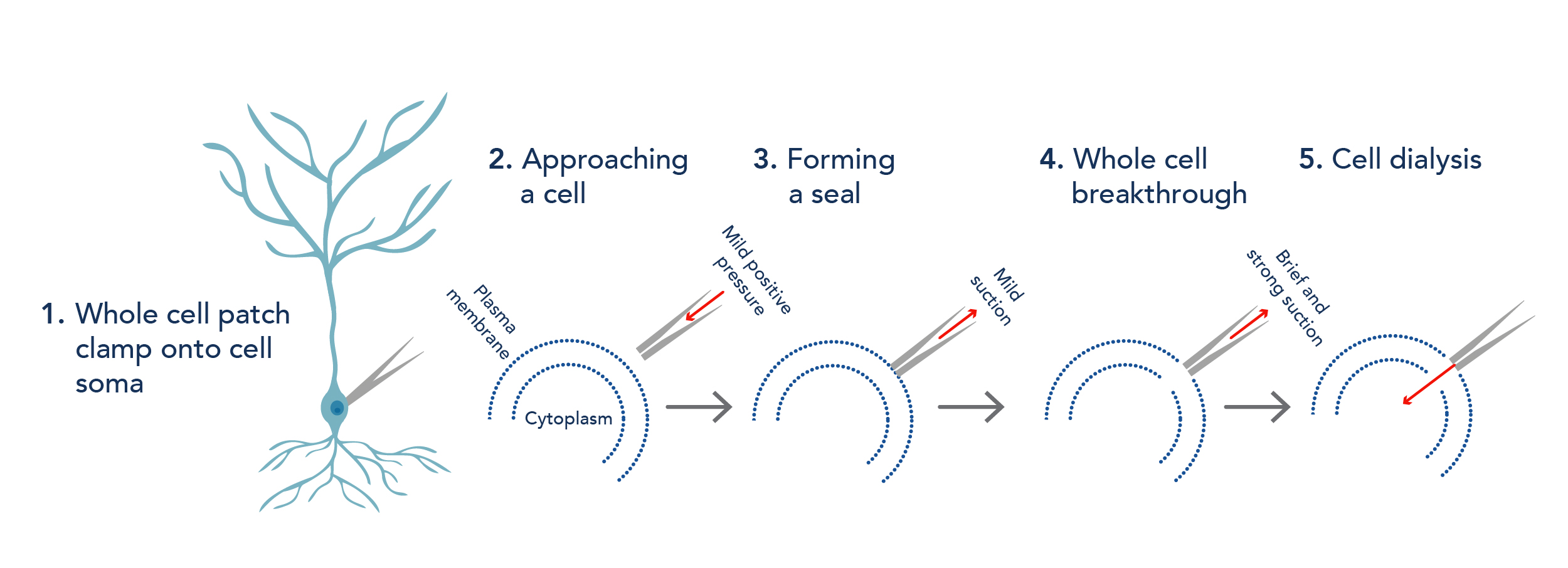
Figure 2. Key steps in whole cell patch clamp for electrophysiology.
Tips for recording from cortical cells
-
Ensure quality preparation of acute brain slices for recordings. This can involve making fresh slicing solution on each experiment day and considering cutting angle to preserve the morphology of cells (for example, the long apical dendrites of pyramidal cells).
-
Patch cells within an optimal depth. This ensures that you are not targeting unhealthy cells in the necrotic layer, which forms due to slicing, or cells that are too deep in the slice and, thus, too difficult to visualize.
-
Pull glass pipettes the day of recording – this helps to prevent dust and other debris from clogging the tip of the pipettes when patching.
-
Maintain your ephys rig! This includes cleaning any spills around the bath chamber, good cable management, and re-chloriding electrodes regularly.
-
Sometimes patching is a numbers game – if you find that the cell you are trying to patch seems unhealthy (e.g. unable to form a seal, very depolarized resting membrane potential, etc.), it is best to move on and target a different cell or use a different slice.
Identifying Cell Types in the Cortex
Electrophysiology properties
One way of identifying a cell type is by looking at their electrophysiology properties in response to a prolonged (~500 ms) current injection during patch clamp. Glia cells can be easily differentiated from neurons by their passive membrane properties and lack of action potentials in response to an influx of positive current. Neuronal cells, on the other hand, can be identified based on their action potential (spiking) patterns.
Pyramidal cells are known to have an accommodating spiking pattern. This means that when a burst of action potentials is elicited by a long current injection, the timing between subsequent action potentials will increase. Other cell types, such as basket cells, are non-accommodating and fast-spiking. Even with these general guides, electrophysiology properties alone are usually not sufficient to determine cell type, because different cell types can have overlapping spiking profiles. For example, both pyramidal cells and Martinotti cells have accommodating spike patterns. Therefore, it is often advised to perform electrophysiology in conjugation with other assays that can help further characterize cell types.
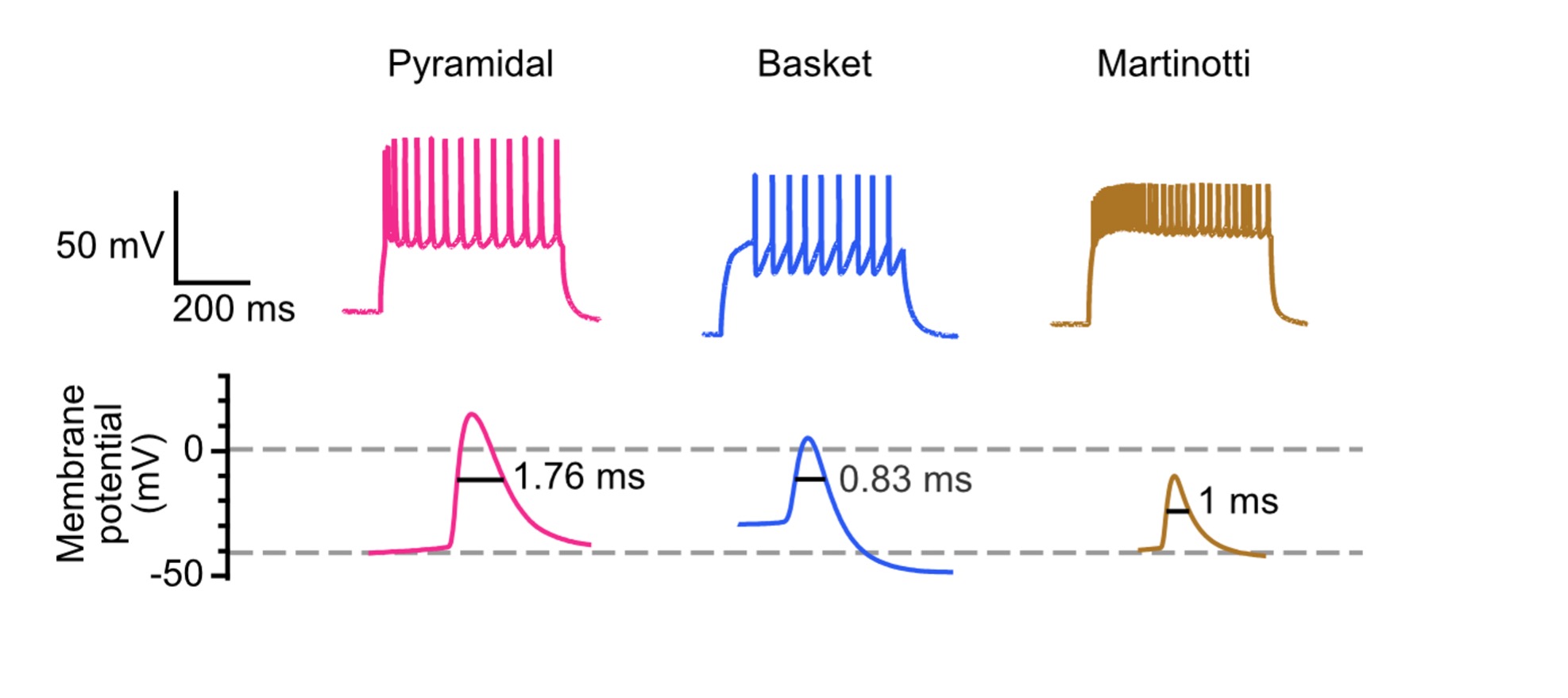
Figure 3. Electrophysiological properties, such as spike pattern and spike half-width, can be used as a guide to identify cortical cell types.
Morphology
Combining morphology with electrophysiology helps researchers to study the function and structure of cells together. Biocytin or biotin can be added into the internal solution of the patch pipette to dialyse patched cells. Streptavidin or avidin conjugated to a fluorescent protein can then be used to target biocytin or biotin. This allows cell morphology to be visualized under, for example, a confocal microscope, which generally gives better resolution than a microscope at an ephys rig. Reconstruction of morphology in 3D from acquired image stacks can also be achieved using tracing software and this can be used to identify cell types.
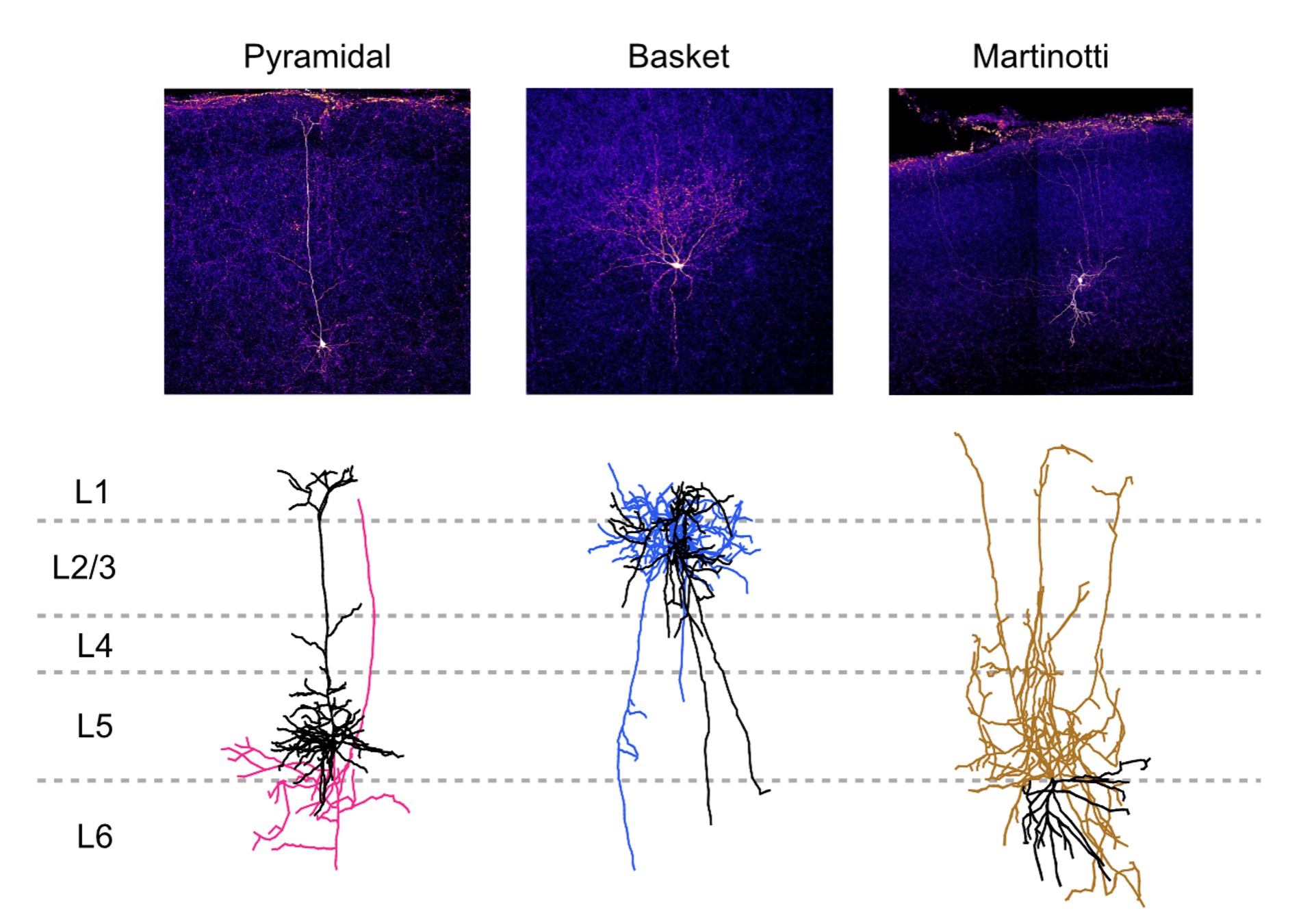
Figure 4. Example morphologies of different cell types after biocytin staining (top) and morphological reconstruction (bottom; black: dendrites, color: axons). Morphology can be used as a further guide to identify cortical cell types. Image credit: Dr. Christina Chou, Cleo Huang, Tal Klimenko, and Vivian Wu from McGill University.
Immunofluorescence staining
Combining immunofluorescence staining for cell type-specific markers can be used to further identify cortical cell types. This can be used on its own or it can be used in tandem with biocytin staining to affirm the cell type of the patched cell.
Different neuronal cell types express certain non-overlapping markers that can be targeted by antibodies. For example, cortical pyramidal cells can be identified with CaMKII. Basket cells are parvalbumin-positive cells, so anti-parvalbumin antibodies can be used to ascertain this cell type. Glia cells also make up the cortex and antibodies can be particularly useful for determining the specific type of glia cell. The table below gives a list of common cortical cell types and their commonly used markers.
|
Cortical Cell Type |
Common Markers |
|
Pyramidal |
|
|
Basket cell |
|
|
Martinotti cell |
|
|
VIP interneuron |
|
|
Astrocytes |
|
|
Microglia |
|
|
Oligodendrocyte |
CNPase, PLP1 |
|
Oligodendrocyte precursor cell |
Table 1. Some common markers used to identify different major cortical cell types.
It is important to note that if immunofluorescence staining is performed on a patched cell, certain cytosolic markers will wash out during whole-cell recordings and immunostaining signal intensity may be weaker. This is important to keep in mind when performing immunostaining after a long patch clamp recording. Parvalbumin, calbindin, calretinin are markers that are commonly affected by this problem.
Summary
The interplay between the various cell types in the cerebral cortex contributes to the proper functioning of cortical circuits. With the tools described above, we can accurately identify cell types and deepen our understanding of neurological conditions. When combining the above tools with even more advanced techniques, such as Cre-Lox recombination and optogenetics, we are also able to study cortical connectivity and function, furthering our understanding of the brain.
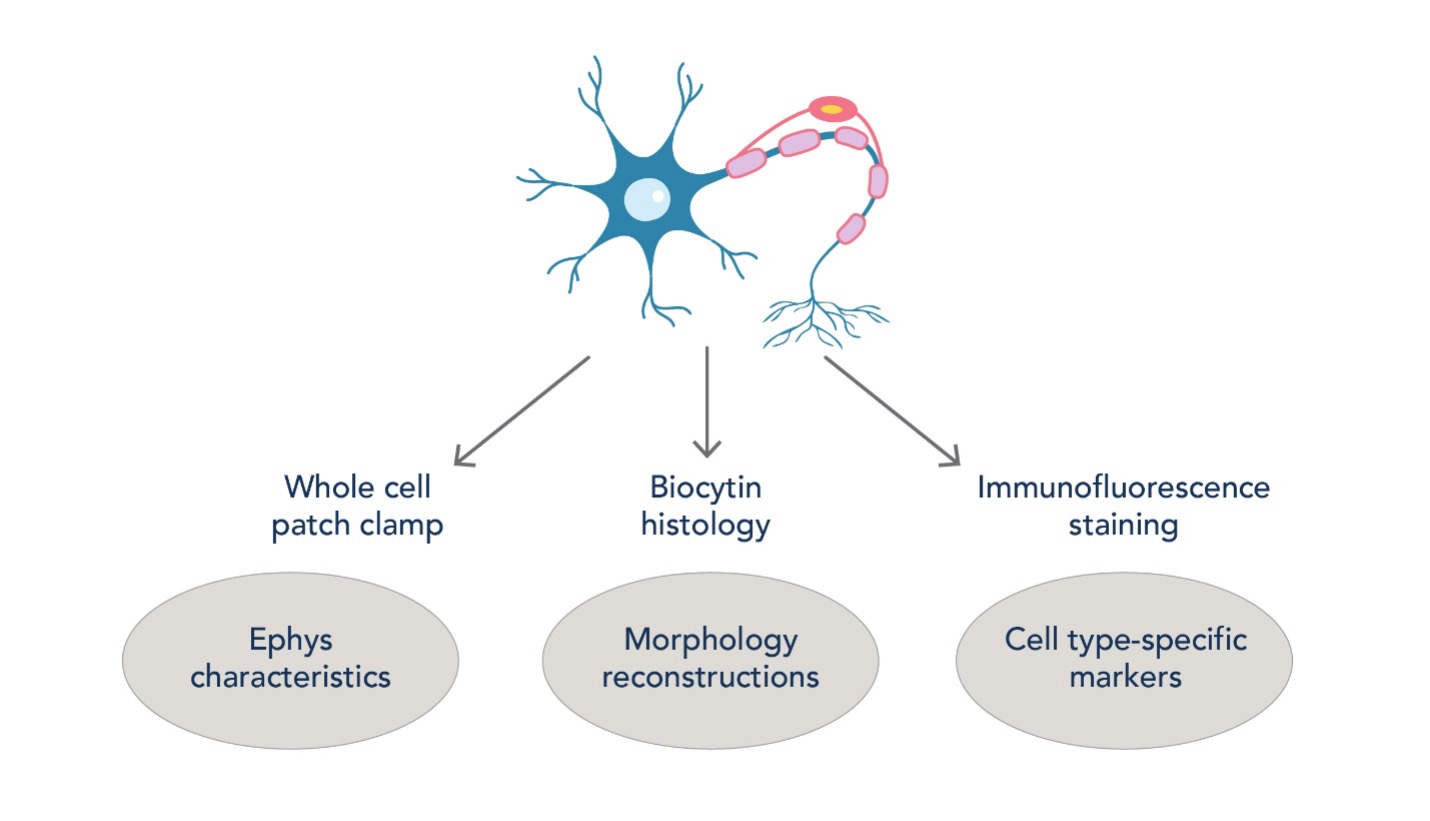
Figure 5. Workflow of identifying cortical cell types using a combination of electrophysiology, morphology, and immunostaining. This arsenal of techniques helps us study cortical microcircuitry with cell type-specific precision.
References
-
Swietek, B., Gupta, A., Proddutur, A., & Santhakumar, V. (2016). Immunostaining of Biocytin-filled and Processed Sections for Neurochemical Markers. Journal of Visualized Experiments: JoVE, (118), 54880. https://doi.org/10.3791/54880
-
Kohara, K., Pignatelli, M., Rivest, A. J., Jung, H. Y., Kitamura, T., Suh, J., Frank, D., Kajikawa, K., Mise, N., Obata, Y., Wickersham, I. R., & Tonegawa, S. (2014). Cell type-specific genetic and optogenetic tools reveal hippocampal CA2 circuits. Nature Neuroscience, 17(2), 269–279. https://doi.org/10.1038/nn.3614
-
Chou, C. Y., Wong, H. H., Guo, C., Boukoulou, K. E., Huang, C., Jannat, J., Klimenko, T., Li, V. Y., Wu. V. C., & Sjöström, P. J. (2025). Principles of visual cortex excitatory microcircuit organization. The Innovation, 6(1), 100735. DOI: 1016/j.xinn.2024.100735
Related Content
The FlexAble way to improve your immunofluorescence with IBEX
A Guide to Immunostaining the Cerebellum
How to choose the right secondary antibody?
Biomarkers for neurodegenerative disorders
Support
Newsletter Signup
Stay up-to-date with our latest news and events. New to Proteintech? Get 10% off your first order when you sign up.

