Validation Data Gallery
Technical Specifications
| GeneID | 7422 |
| Species | Human |
| Expression | HEK293 |
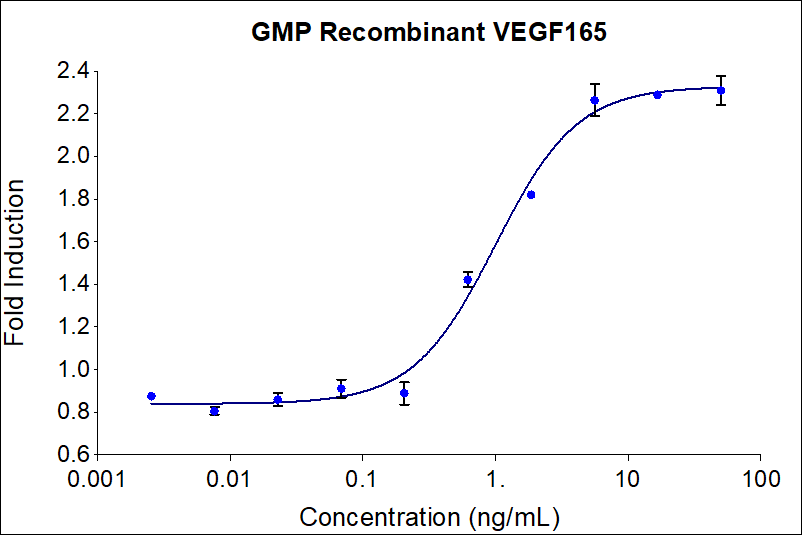
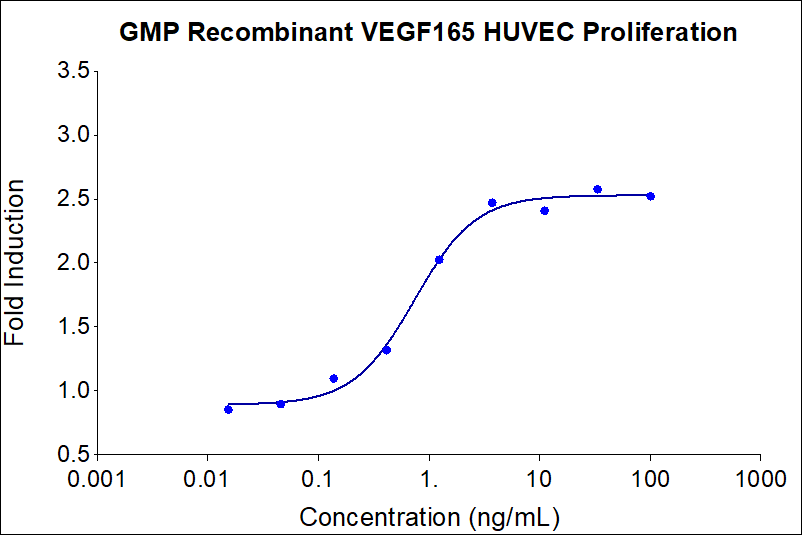
| EC50 | 0.3-3.75 ng/mL |
| Activity(Secondary) | 0.3-3.75 ng/mL |
| Purity | >95% |
| Endotoxin | < 0.1 EU/µg |
| Accession Number | P15692-4 |
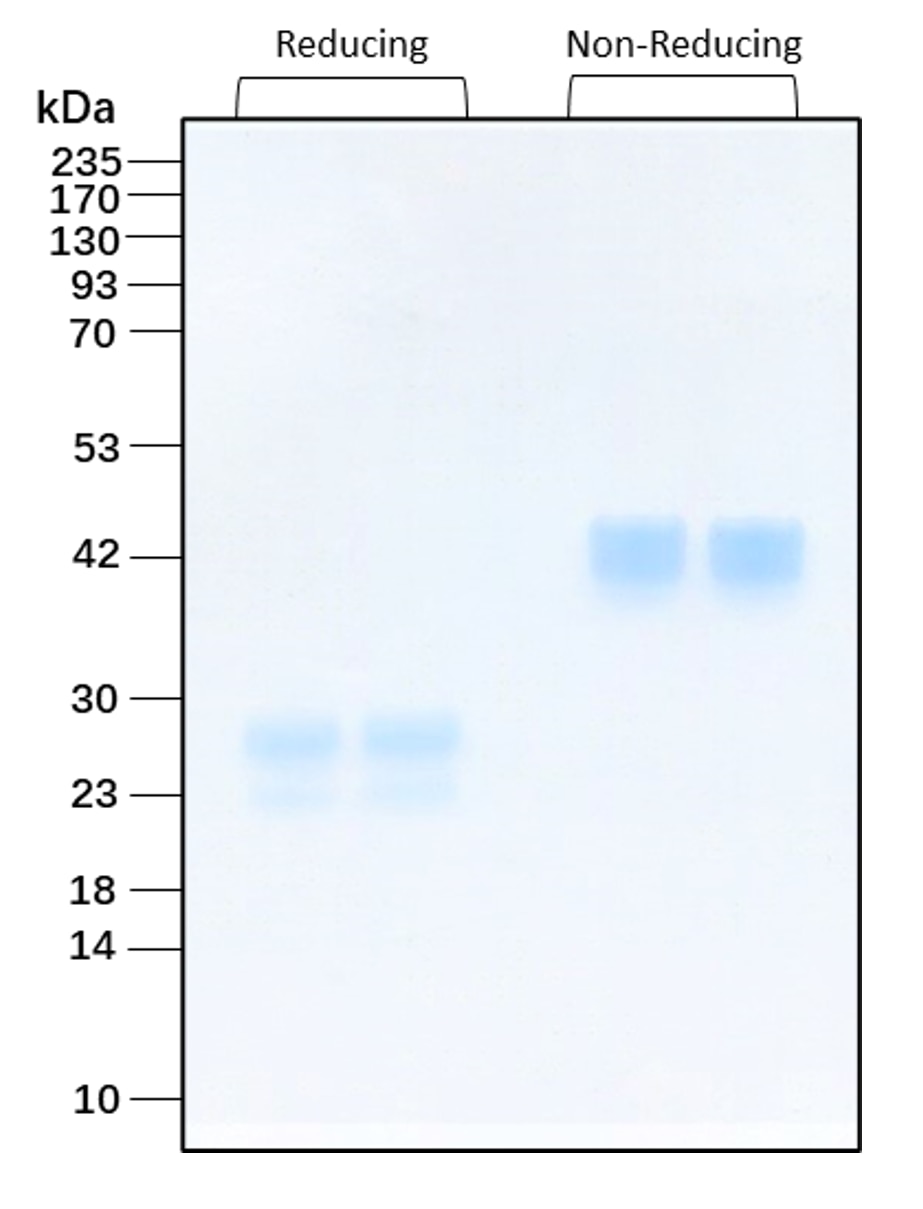
| Molecular Mass | 22 to 30 and 50 kDa reduced, 40 to 50 and 75 kDa non-reduced, homodimer, glycosylated |
| Formulation | 1x PBS, See Certificate of Analysis for details |
| Species Reactivity | human |
Stability and Reconstitution
| Stability and Storage | Product Form | Temperature Conditions | Storage Time (From Date of Receipt) |
|---|---|---|---|
| Lyophilized | -20°C to -80°C | Until Expiry Date | |
| Lyophilized | Room Temperature | 2 weeks | |
| Reconstituted as per CofA | -20°C to -80°C | 6 months | |
| Reconstituted as per CofA | 4°C | 1 week | |
| Avoid repeated freeze-thaw cycles. | |||
| Reconstitution | Briefly centrifuge the vial before opening. It is recommended to reconstitute the protein to 0.2 mg/mL in sterile 1xPBS pH 7.4 containing 0.1% endotoxin-free recombinant serum albumin (HSA). Gently swirl or tap vial to mix. |
GMP Quality Policies
HumanKine® GMP (Good Manufacturing Practice) recombinant proteins are manufactured and validated in accordance with ISO 13485 quality management system and is compliant with GMP.
Our GMP recombinant proteins are animal component free (ACF), xeno free (XF) and tag free (TF). Read more about these policies here.
Background
This gene is a member of the PDGF/VEGF growth factor family. It encodes a heparin-binding protein, which exists as a disulfide-linked homodimer (PMID 10838264; 11329058). This growth factor induces proliferation and migration of vascular endothelial cells and is essential for physiological and pathological angiogenesis. Misregulations of this gene in mice result in abnormal embryonic blood vessel formation.
Synonyms
L VEGFA, MVCD1, Vascular permeability factor, VEGF, VEGF A, VEGF165, VEGFA, VPF
Publications
| Species | Title |
|---|---|
Neural Regen Res A matrix metalloproteinase-responsive hydrogel system controls angiogenic peptide release for repair of cerebral ischemia/reperfusion injury |
Reviews
The reviews below have been submitted by verified Proteintech customers who received an incentive for providing their feedback.
FH Xunyu (Verified Customer) (07-26-2022) | great product, easy to use.
|