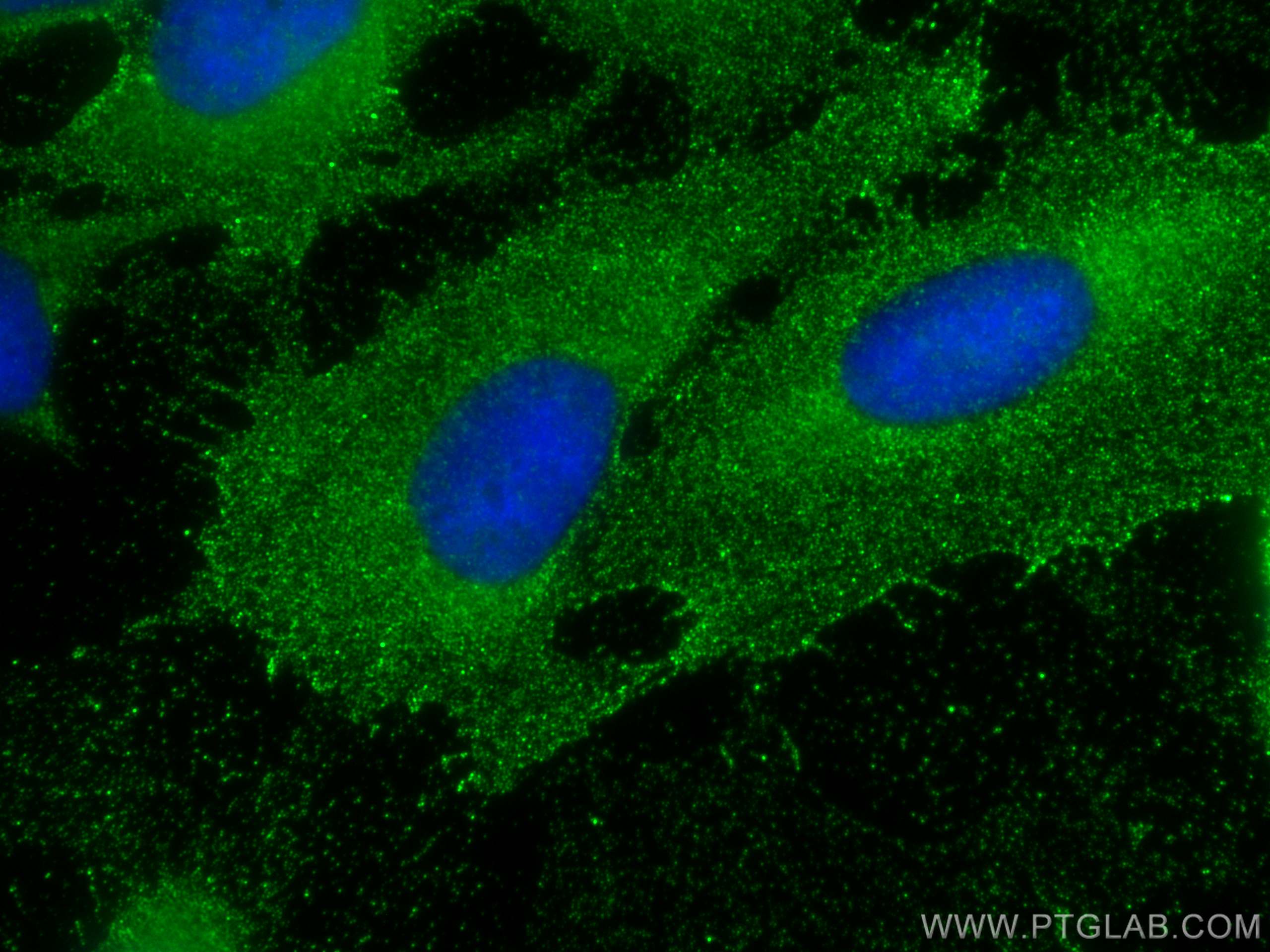
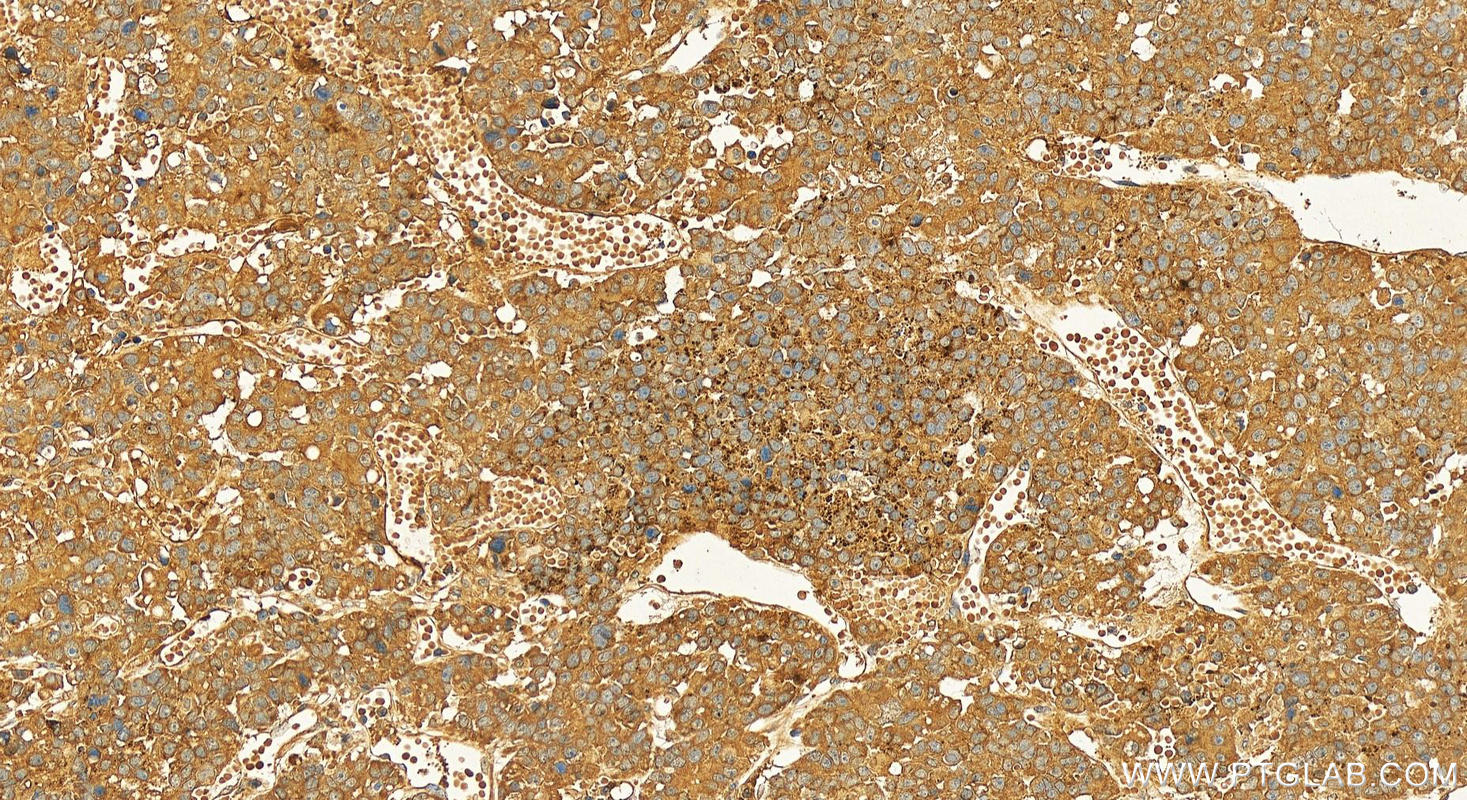
Tested Applications
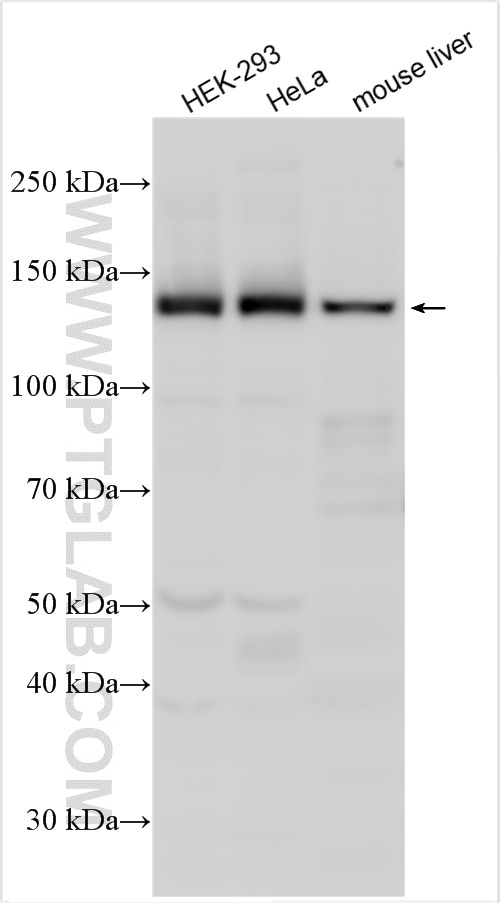
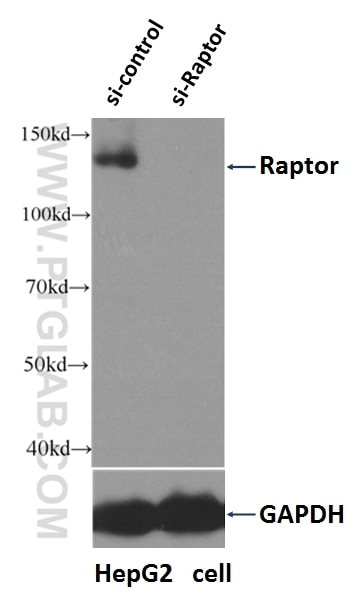
| Positive WB detected in | HEK-293 cells, HepG2 cells, HeLa cells, mouse liver tissue |
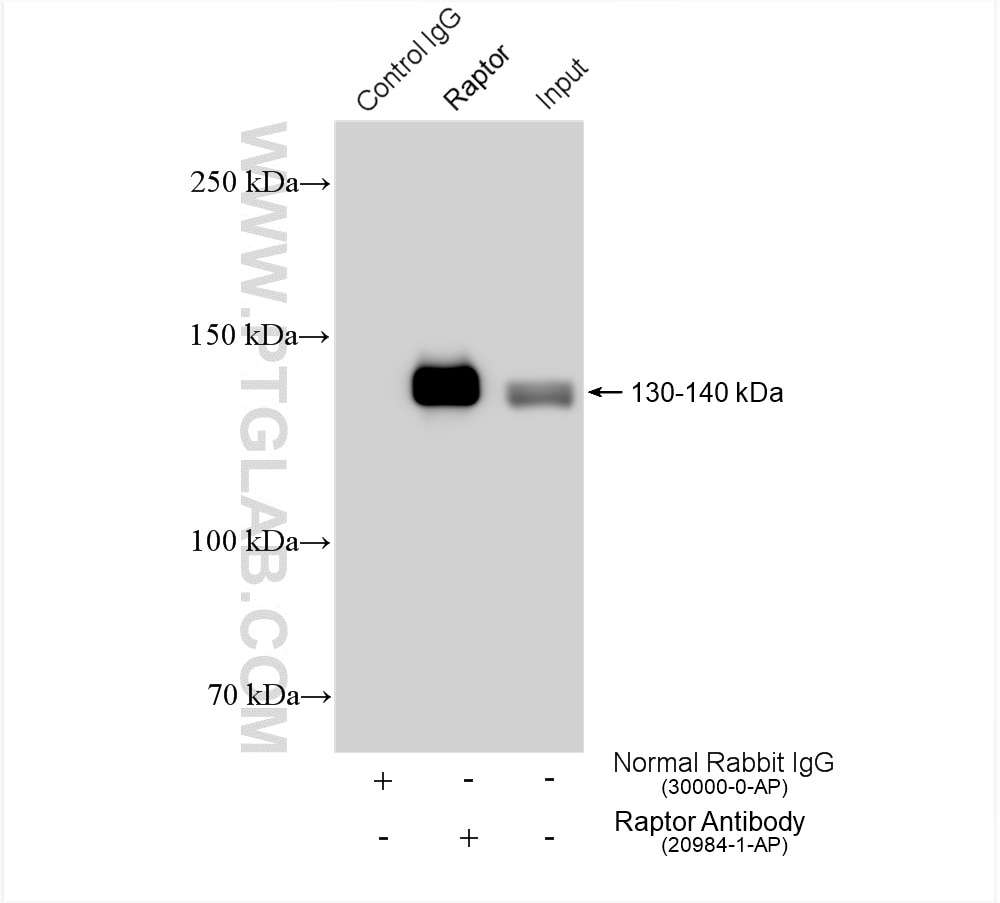
| Positive IP detected in | HEK-293 cells |
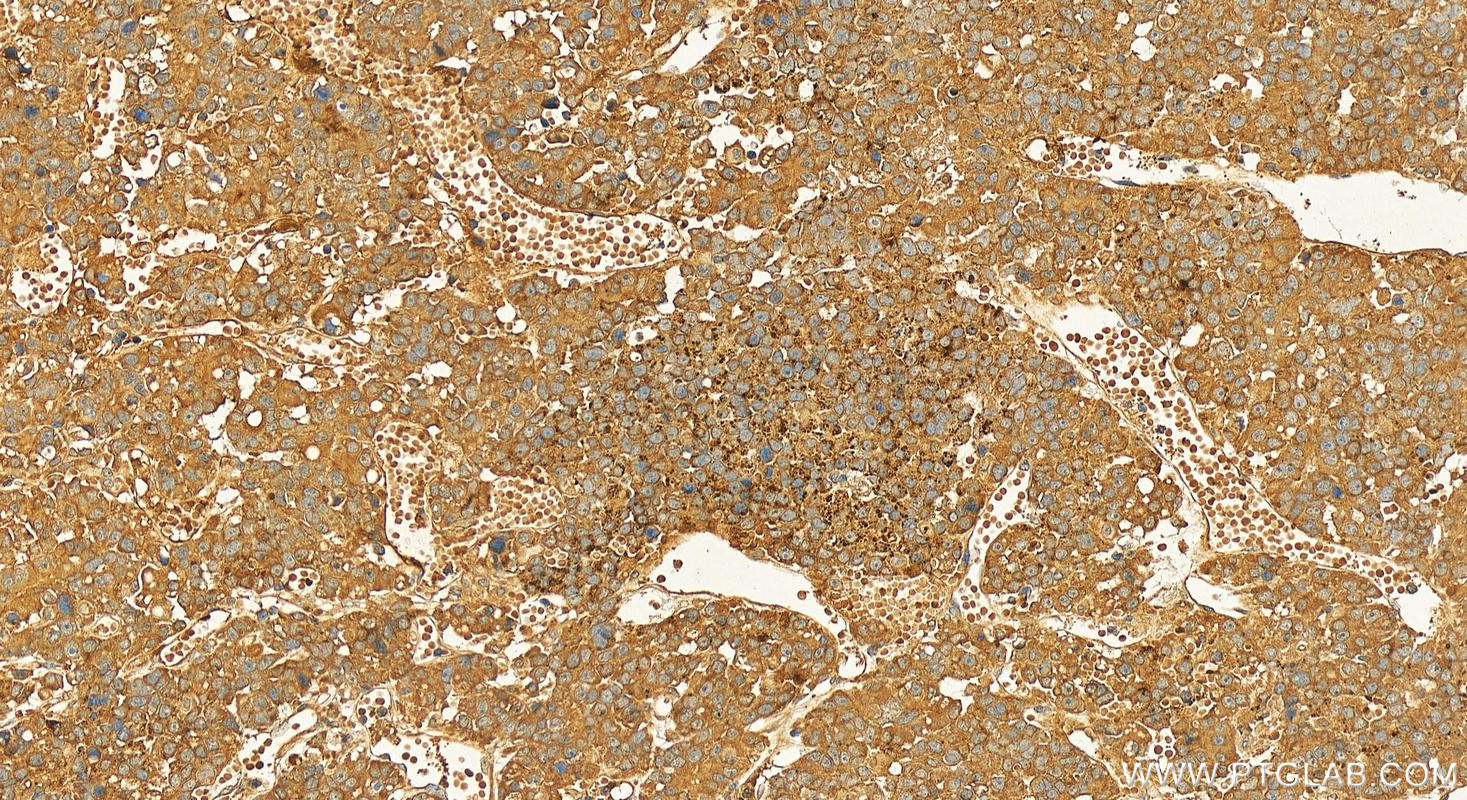
| Positive IHC detected in | human hepatocellular carcinoma Note: suggested antigen retrieval with TE buffer pH 9.0; (*) Alternatively, antigen retrieval may be performed with citrate buffer pH 6.0 |
| Positive IF/ICC detected in | U2OS cells |
Recommended dilution
| Application | Dilution |
|---|---|
| Western Blot (WB) | WB : 1:2000-1:12000 |
| Immunoprecipitation (IP) | IP : 0.5-4.0 ug for 1.0-3.0 mg of total protein lysate |
| Immunohistochemistry (IHC) | IHC : 1:50-1:500 |
| Immunofluorescence (IF)/ICC | IF/ICC : 1:50-1:500 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
Published Applications
| KD/KO | See 1 publications below |
| WB | See 42 publications below |
| IHC | See 2 publications below |
| IF | See 12 publications below |
| IP | See 3 publications below |
Product Information
20984-1-AP targets Raptor in WB, IHC, IF/ICC, IP, ELISA, PLA applications and shows reactivity with human, mouse samples.
| Tested Reactivity | human, mouse |
| Cited Reactivity | human, mouse, rat, rabbit |
| Host / Isotype | Rabbit / IgG |
| Class | Polyclonal |
| Type | Antibody |
| Immunogen |
Peptide Predict reactive species |
| Full Name | raptor |
| Calculated Molecular Weight | 149 kDa |
| Observed Molecular Weight | 130-150 kDa |
| GenBank Accession Number | NM_020761 |
| Gene Symbol | Raptor |
| Gene ID (NCBI) | 57521 |
| RRID | AB_11182390 |
| Conjugate | Unconjugated |
| Form | Liquid |
| Purification Method | Antigen affinity purification |
| UNIPROT ID | Q8N122 |
| Storage Buffer | PBS with 0.02% sodium azide and 50% glycerol, pH 7.3. |
| Storage Conditions | Store at -20°C. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. 20ul sizes contain 0.1% BSA. |
Background Information
RPTOR, also named as KIAA1303 and RAPTOR Belongs to the WD repeat RAPTOR family. It is involved in the control of the mammalian target of rapamycin complex 1 (mTORC1) activity which regulates cell growth and survival, and autophagy in response to nutrient and hormonal signals; functions as a scaffold for recruiting mTORC1 substrates. mTORC1 is activated in response to growth factors or amino-acids. Amino-acid-signaling to mTORC1 is mediated by Rag GTPases, which cause amino-acid-induced relocalization of mTOR within the endomembrane system. Activated mTORC1 up-regulates protein synthesis by phosphorylating key regulators of mRNA translation and ribosome synthesis. mTORC1 phosphorylates EIF4EBP1 and releases it from inhibiting the elongation initiation factor 4E (eiF4E). mTORC1 phosphorylates and activates S6K1 at 'Thr-389', which then promotes protein synthesis by phosphorylating PDCD4 and targeting it for degradation. The antibody is specific to RPTOR.
Protocols
| Product Specific Protocols | |
|---|---|
| IF protocol for Raptor antibody 20984-1-AP | Download protocol |
| IHC protocol for Raptor antibody 20984-1-AP | Download protocol |
| IP protocol for Raptor antibody 20984-1-AP | Download protocol |
| WB protocol for Raptor antibody 20984-1-AP | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
Publications
| Species | Application | Title |
|---|---|---|
Mol Cell Acetyl-CoA Derived from Hepatic Peroxisomal β-Oxidation Inhibits Autophagy and Promotes Steatosis via mTORC1 Activation. | ||
Autophagy Buddleoside alleviates nonalcoholic steatohepatitis by targeting the AMPK-TFEB signaling pathway | ||
Nat Commun N7-methylguanosine tRNA modification promotes esophageal squamous cell carcinoma tumorigenesis via the RPTOR/ULK1/autophagy axis. | ||
Nat Commun Location-specific inhibition of Akt reveals regulation of mTORC1 activity in the nucleus. |